Past Issues
High Throughput Sequencing Detect Gingivitis and Periodontal Oral Bacteria in Alzheimer’s Disease Autopsy Brains
Huma Siddiqui1, Emenike RK Eribe1, Sim K Singhrao2*, Ingar Olsen1
1Department of Oral Biology, Faculty of Dentistry, University of Oslo, Norway 2Dementia & Neurodegenerative Diseases Research Group, Faculty of Clinical and Biomedical Sciences, School of Dentistry, University of Central Lancashire, Preston, United Kingdom
*Corresponding Author: Sim K Singhrao, Dementia & Neurodegenerative Diseases Research Group, Faculty of Clinical and Biomedical Sciences, School of Dentistry, University of Central Lancashire, Preston, United Kingdom.
Received: Aug 17, 2019 Published: Sept 10, 2019
ABSTRACT
The sporadic form of Alzheimer’s disease (AD) is the most common form of dementia characterized microscopically by the presence of amyloid-beta (Aβ) plaques and tau neurofibrillary tangles with the clinical presentation of cognitive deficit. Its etiology remains obscure. The cited literature from epidemiological studies suggests the presence of gingivitis and periodontitis almost double the risk for AD over a 10-year period. This feasibility study used 16S rDNA high throughput sequencing to evaluate the bacterial components of the oral microbiome in snap-frozen human AD and non-AD control brains. Specimens (n=20), 10 from AD brains and 10 from non-AD age-matched brains were subjected to molecular barcoding “blindly” with high throughput sequencing. Initial PCR treatment, using 14 different primer sets separately and in combination, identified 4 (40%) positive samples in the AD-group and 6 (60%) positive samples in the non-ADgroups. with bacterial species associated with the oral and the gastrointestinal tract. Brain cell lysates were validated for the presence of bacterial peptidoglycan and showed 8 out of 10 brains to be positive for this universal bacterial protein in both the AD and non-AD groups. Actinomycetales and Prevotella (bacterial marker for gingivitis) and Treponema and Veillonella, (periodontitis) were present exclusively in the AD group.This study confirmed Actinomycetales and Bacteroidales (Treponema and Veillonella species) were exclusively isolated from AD brain tissue, and supports other epidemiological studies which demonstrate gingivitis and periodontal disease to be associated with AD.
KEYWORDS: Alzheimer’s Disease; Human Brains; High Throughput Sequencing; 16S rDNA; Bacteria; Oral; Intestinal.
INTRODUCTION
Alzheimer’s disease (AD) is a neurodegenerative disease and represents the most common form of dementia. Its prevalence is still rising worldwide [1]. Atrophy and neuronal death in the frontotemporal region including the hippocampus, containing amyloid-beta plaques and neurofibrillary tangles, together with a clinical history of dementia confirm presence of AD. The clear majority suffers from the sporadic form of AD (95% of the cases) [2] in which interplay between both susceptibility genes and environmental factors is implicated [3]. One of the greatest risk factors for developing AD is advancing age, which also encompasses an aging immune system that has the potential to facilitate dysbiosis of host microbiomes.
The apolipoprotein ApoE ε4 polymorphic isoform is common to all forms of AD [4]. Patients carrying the ApoE4 gene suffer from hyperlipidaemia and this predisposes them to atherosclerosis [5], bacterial infections [6,7] and the ensuing inflammation [7,8]. The ApoE4 susceptibility gene acts in combination with environmental risk factors to cause pathology. These concepts have given rise to the importance of identifying modifiable risk factors together with management of these risk factors in order to either delay or prevent the onset of dementia. For example, dysbiotic oral and gastrointestinal tract host microbiome(s) represent a potential environmental risk factor. Pathogens may spread from their primary niche to the brain, encouraging AD development [2, 9,10]. In addition, epidemiological studies identified a 10 year exposure to both gingivitis and periodontal disease promoted AD pathogenesis [11,12]. Furthermore, a prospective laboratory-based study demonstrated the directionality and the timeline of periodontal disease being a risk for AD by showing circulating antibodies to Fusobacterium nucleatum and Prevotella intermedia (oral bacteria) to be associated to a cognitive deficit 10 years later [13]. Notably Stein et al., [14] showed tooth loss due to periodontal disease can almost double the risk for AD onset.
The evidence supporting that inheritance of the ApoE4 allele being an inflammatory phenotype is demonstrated experimentally via an increased secretion of inflammatory cytokines [8] and an excessively activated complement cascade [15,16]. Inflammatory mediators increase the permeability of the blood-brain barrier (BBB) to circulating pathogens, serum proteins and cytokines in the systemic circulation, providing an opportunity for the influx of dysbiotic bacteria into the brain [8,17-19]. This is illustrated by the susceptibility of the ApoE4 carriers to Chlamydia pneumoniae and herpes simplex virus (HSV) type 1 infections in AD brains [20-22]. Furthermore, those individuals having inherited the ApoE4 polymorphic gene [23] demonstrate Chlamydia-associated infections are able to reach the brain via infected monocytes following increased BBB permeability [24]. For a more focused review on this subject see ref [25].
Although a polymicrobial component is an essential contributor to periodontal pathogenesis, this does not completely exclude other non-microbial related factor(s) such as lifestyle behaviours (poor oral hygiene, smoking, excessive drinking, stress) and host’s immune responses. A variety of microorganisms including C. pneumoniae [23], HSV type 1 [26] and periodontal pathogens, particularly Porphyromonas gingivalis (previously Bacteroides gingivalis) and Treponema species have been detected in AD brains [27-30], but their relevance to the pathogenesis of this neurodegenerative disease, is debated. However, due to the recent in vivo findings of oral infections of rodents with P. gingivalis [17], and its association with Actinobacteria in AD brains argues at least some relevance for the pathogenesis of AD in the context of bacterial infections.
Currently there is only one published report that analysed the bacterial component of the host’s microbiome in frozen and fixed post-mortem tissue from AD and control cases [31]. This initial study [31] used the 16S rDNA approach for its sensitivity and suitability to analyze bacterial DNA in frozen and fixed brain tissue.
The current study used high throughput sequencing and molecular barcoding to investigate the presence of bacterial genera in the same cohort of AD brain tissues that were examined in a previous study using biochemical and immunofluorescence methodology that detected lipopolysaccharide (LPS) as a signature of P. gingivalis in AD but not control cases [9].
MATERIALS AND METHODS
Bacterial DNA evaluation
Brain tissue samples: The human brain tissue used in this study was obtained from the Brains for Dementia Research (BDR) network and was kindly provided by the Newcastle Brain Tissue Resource in 2010 for a PhD project [9,32]. A new request was approved by the BDR for re-use of surplus human brain tissue samples for this study. All research procedures met approval of our academic institute (University of Central Lancashire, Ref. No. 343) and the ethical guidelines, including adherence to the legal requirements of the study in the UK. The study also received regional approval from the Norwegian REC (Regional Committee for Medical and Health Research Ethics, Southeast, Ref. No. 20152386).
The specimens were obtained from neuropathologically confirmed AD (n=10) patients comprising of eight females and two male donors with average age (79 and 83 years) respectively (Table 1). Where possible, age-matched non-AD control (n=10) brains, seven females and three males with average age (84 and 73 years respectively) were used. Samples of frozen human brain tissue were taken from an area of the brain adjacent to the lateral ventricle of the parietal lobe. On receipt, all specimens were allocated a code number and thereafter all data recorded from those cases were identified by that code. The cases are identified here as AD and non-AD controls for clarity of reporting. Related information (age, gender, cause of death, ApoE allelic form, post-mortem and storage interval, number of bacteria detected by high throughput sequencing compared with the laboratory PCR methodology) for each anonymized case is given in Table 1.
Table 1: The age, post-mortem interval/cause of death, storage interval, and cases in which bacterial DNA detected in the cases analysed.
|
Case |
Age and sex |
Post-mortem interval (hours) |
Cause of death and ApoE allelic form (in bold text) |
Storage interval in years |
Bacterial numbers detected by high throughput sequencing |
Bacterial DNA detected by lab PCR |
|
AD 1 |
78/F |
12 |
Unknown/unknown |
21 |
0 |
Yes |
|
AD 2 |
77/F |
8 |
Unknown/unknown |
19 |
19,421 |
Yes |
|
AD 3* |
84/F |
8 |
1a heart failure due to 1b senile dementia. 3/4 |
19 |
1,266 |
No |
|
AD 4 |
84/F |
8 |
likely bronchopneumonia. 3/3 |
19 |
0 |
No |
|
AD 5* |
85/F |
9 |
Unknown. 3/3 |
18 |
0 |
No |
|
AD 6 |
83/M |
9 |
Unknown. 4/4 |
17 |
0 |
No |
|
AD 7 |
80/F |
4 |
pulmonary embolism. 4/4 |
17 |
0 |
Yes |
|
AD 8* |
83/F |
10 |
unknown/unknown |
16 |
2,573 |
Yes |
|
AD 9 |
63/F |
11 |
Bronchopneumonia. 3/4 |
9 |
0 |
Yes |
|
AD 10* |
83/M |
12 |
Unknown. 2/2 |
5 |
5,374 |
Yes |
|
Non-AD 1 |
69/F |
16 |
gastric cancer. 3/3 |
9 |
503 |
Yes |
|
Non-AD 2 |
72/M |
17 |
esophageal adenocarcinoma. 3/4 |
9 |
0 |
Yes |
|
Non-AD 3 |
103/F |
21 |
Pneumonia. 3/3 |
9 |
40,045 |
No |
|
Non-AD 4 |
78/F |
23 |
metastatic esophageal carcinoma with broncho-esophageal fistula. 2/3 |
8 |
2,726 |
No |
|
Non-AD 5 |
89/F |
24 |
Heart failure 1a heart failure 1b mixed aortic valve. 3/3 |
8 |
0 |
No |
|
Non-AD 6 |
67/M |
43 |
metastatic laryngeal carcinoma (non-occupational)/unknown |
4 |
0 |
No |
|
Non-AD 7 |
81/M |
34 |
1a pneumonia 1b: infective endocarditis. 2/3 |
3 |
1,670 |
No |
|
Non-AD 8 |
88/F |
34 |
1a aspiration pneumonia, 1b RT total anterior circulation stroke. 3/3 |
4 |
7,795 |
No |
|
Non-AD 9 |
78/F |
22 |
Metastatic cancer, primary origin unknown (probably ovarian). 2/3 |
3 |
0 |
No |
|
Non-AD 10 |
89/F |
22 |
Ischemic bowel and multi-organ failure. 3/3 |
3 |
3,328 |
No |
|
Total bacteria after all filtration = 84.701; * Cases positive for P. gingivalis LPS [9] |
||||||
The remaining tissue samples (approximating to 50 mg of tissue for each brain) were dissected, and genomic DNA isolation was performed in a regularly serviced PCR hood as expected per forensic DNA testing procedures. Brain specimens were thawed out in a PCR hood, and the remaining snap-frozen unfixed tissue was subjected to genomic DNA isolation at the University of Central Lancashire as described below. Each specimen was placed into a sterile labelled Eppendorf® tube containing 180 µl enzymatic buffer (20 mM Tris-Cl, pH 8.0, 2 mM sodium EDTA, and 1.2% triton x-100) to which lysozyme (Sigma-Aldrich Poole, Dorset, UK) was added (20 mg/ml). All lids were closed and then covered with Parafilm® M (Sigma-Aldrich). Following an overnight incubation at 37ºC with shaking, fresh 180 µl of buffer with 20 µl proteinase K (Sigma-Aldrich) were added to each tube and the content was mixed and re-incubated overnight at 56ºC as before. Next day, the tubes from the incubator were removed and an equal volume of phenol/chloroform/isoamyl alcohol (25:24:1) from Sigma-Aldrich was added to the digested sample and vortex-mixed. The tubes were centrifuged at room temperature for 5 min at 21,000 RCF. The upper aqueous phase was collected into a pre-labelled sterile, Eppendorf® tube. A cocktail (glycogen, Sigma-Aldrich, 20 µg/µl, 7.5 M ammonium acetate (Sigma-Aldrich) at pH 5.2 (0.5 × volume of sample) and 100% ethanol, 2.5 × volume of sample) were added, reagent by reagent, into the aqueous phase. The tubes were placed at -20ºC overnight to precipitate DNA. Following precipitation, the tubes containing each sample were centrifuged at 4ºC for 30 min at 21,000 RCF to pellet the genomic DNA. The pellets were washed three times in 150 µl 70% ice cool ethanol and centrifuged twice at 4ºC for 2 min at 21,000 RCF. The isolated genomic DNA (in ethanol) was sent to the University of Oslo, Oslo, Norway via an overnight courier service. All specimens were coded to ensure the experimenter remained completely unaware of which cases corresponded to AD and non-AD control brains.
PCR and high throughput sequencing
At the University of Oslo, DNA samples (n=20) were suspended in 100 µl Qiagen buffer (Qiagen Instruments AG, Stockach, Germany) and subjected to PCR using 14 different primer sets (Eurofins Genomics, Ebersberg, Germany) (Table 2). Combinations of forward and reverse primers targeting the V4 region were used: 502F/701R, 503F/701R, 501F/702R, 503F/702R, 504F/702R, 507F/702F, 508F/702R, 503F/703R, 504R/703R, and 507F/703R [33]. The v3F.Forsyth primer was combined with 701R-708R. PCR reactions were performed (in triplicate) for each sample. Each 20 μl volume of PCR reaction contained the following (1 μl of 10 pool/μl forward and reverse primers, 2 μl of template DNA and 16 μl AccuPrime™ Pfx SuperMix mixture (Thermo Fisher Scientific, Wilmington, DE) consisting of 22 U/ml Thermococcus species KOD thermostable polymerase complexed with anti-KOD antibodies (Invitrogen, Carlsbad, CA), 66 mM Tris-SO4 (pH 8.4), 30.8 mM (NH4)2SO4, 11 mM KCl, 1.1 mM MgSO4, 330 µM dNTPs, AccuPrime™ proteins, and stabilizers purchased from Invitrogen). Prior to PCR, pipettes, tips and Eppendorf® tubes were decontaminated under UV irradiation for 30 min in the PCR assembling hood. PCR amplifications were performed on a GeneAmp PCR System 2700 instrument (Applied Biosystems) using sterile reagents with initial denaturation at 95ºC for 5 min followed by 30 cycles of denaturation at 95ºC for 20 s, annealing at 55ºC for 30 s, extension at 72ºC for 60 s, with a final extension at 72ºC for 10 min. The product was stored at 4ºC. Replicate amplicons were pooled and visualized on 1.0 % Seakam agarose gels using 10 μl GelRed Nucleic acid binding dye (Biotium Inc., Fremont, CA) in 1 × TBE buffer. The amplicons were cleaned using a SequalPrepTM Normalization Plate (96) kit (Invitrogen) according to the manufacturer’s instructions. Molecular identifier (MID) tags, 10-mer, were used as sample identifiers (Table 2). All pooled PCR products were purified using the Agencourt AMPure PCR purification system (Beckman Coulter, Brea, CA). The recovered DNA quantity was recorded (Nanodrop 3300 Flurospectrometer, Thermo Fisher Scientific).
Table 2: PCR primers used in this study.
|
No. |
Oligo name |
Sequence |
|
1 |
V4.SA501 |
5’-AATGATACGGCGACCACCGAGATCTACACATCGTACGTATGGTAATTGTGTGCCAGCMGCCGCGGTAA -3’ |
|
2 |
V4.SA502 |
5’-AATGATACGGCGACCACCGAGATCTACACACTATCTGTATGGTAATTGTGTGCCAGCMGCCGCGGTAA -3’ |
|
V4.SA503 |
5’-AATGATACGGCGACCACCGAGATCTACACTAGCGAGTTATGGTAATTGTGTGCCAGCMGCCGCGGTAA-3’ |
|
|
3 |
V4.SA504 |
5’-AATGATACGGCGACCACCGAGATCTACACCTGCGTGTTATGGTAATTGTGTGCCAGCMGCCGCGGTAA-3’ |
|
4 |
V4.SA507 |
5’-AATGATACGGCGACCACCGAGATCTACACGGATATCTTATGGTAATTGTGTGCCAGCMGCCGCGGTAA -3’ |
|
5 |
V4.SA508 |
5’-AATGATACGGCGACCACCGAGATCTACACGACACCGTTATGGTAATTGTGTGCCAGCMGCCGCGGTAA -3’ |
|
6 |
V4.SA701 |
5’-CAAGCAGAAGACGGCATACGAGATAACTCTCGAGTCAGTCAGCCGGACTACHVGGGTWTCTAAT -3’ |
|
7 |
V4.SA702 |
5’-CAAGCAGAAGACGGCATACGAGATACTATGTCAGTCAGTCAGCCGGACTACHVGGGTWTCTAAT-3’ |
|
8 |
V4.SA703 |
5’-CAAGCAGAAGACGGCATACGAGATAGTAGCGTAGTCAGTCAGCCGGACTACHVGGGTWTCTAAT-3’ |
|
9 |
V4.SA704 |
5’-CAAGCAGAAGACGGCATACGAGATCAGTGAGTAGTCAGTCAGCCGGACTACHVGGGTWTCTAAT-3’ |
|
10 |
V4.SA705 |
5’-CAAGCAGAAGACGGCATACGAGATCGTACTCAAGTCAGTCAGCCGGACTACHVGGGTW TCTAAT-3’ |
|
11 |
V4.SA706 |
5’-CAAGCAGAAGACGGCATACGAGATCTACGCAGAGTCAGTCAGCCGGACTACHVGGGTWTCTAAT-3’ |
|
12 |
V4.SA707 |
5’-CAAGCAGAAGACGGCATACGAGATGGAGACTAAGTCAGTCAGCCGGACTACHVGGGTWTCTAAT-3’ |
|
13 |
V4.SA708 |
5’-CAAGCAGAAGACGGCATACGAGATGTCGCTCGAGTCAGTCAGCCGG ACTACHVGGGTWTCTAAT-3’ |
|
14 |
V3F.forsyth |
5’-AATGATACGGCGACCACCGAGATCTACACTATGGTAATTGTCCTACGGGAGGCAGCAG-3’ |
High throughput sequencing was performed using the Illumina MiSeq 500-cycle v2 (2 × 250 bp) kit (Illumina, San Diego, CA), which resulted in 15 GB, 25 M SE/50M PE raw reads. Sequencing was performed at the Department of Biochemistry, DNA Sequencing Facility, University of Cambridge, UK (http://www.bioc.cam.ac.uk/dnasequencing). Processing of the sequencing data and taxonomy assignment were accomplished with an algorithm modified and based on that described by Siddiqui et al., [34]. To maximize the assignment rate, raw reads were used directly without quality filtering. Reads were first assigned with sample IDs based on the MID sequences and then BLASTN-searched against a combined set of 16S rRNA reference sequences consisting of the HOMDEXTGG set published by Siddiqui et al., [34] and the NCBI 16S rRNA reference sequence set (ftp://ftp.ncbi.nim.nih.gov/blast/db/16SMicrobial.tar.gz). These combined, well curated and near full-length reference sequences represented 1,151 oral and 12,013 non-oral microbial species.
Statistical analyses
To analyze any significant differences between the microbiota of AD and control brains a statistical method introduced in Metastats (www.metastats.cbcb.umd.edu) was used. This method employs a false discovery rate to improve specificity in high complexity environments, and in addition handles sparsely sampled features using Fisher’s exact test [35]. The Shapiro-Wilk test was used for assessing differences in the post-mortem interval and the non-parametric Mann-Whitney U test for independent samples with IMB SPSS statistical package version 23. A p-value of ? 0.05 was considered significant.
Bacterial peptidoglycan protein validation
Tissue lysates of all AD and non-AD brains: At the University of Central Lancashire, UK, tissue lysates were prepared from all human brains as previously reported [9] and were used for dot blots with the anti-bacterial peptidoglycan antibody (MAB995) for overall presence of bacteria in the specimens.
Dot blot: To confirm the presence of bacterial peptidoglycan, dot blots were performed by transferring 30 µg of total protein onto a polyvinylidene difluoride membrane (PVDF, Immobilon-P, Millipore, UK), which had been previously permeabilized with methanol and hydrated in 1 × transfer buffer (diluted from 10 × stock transfer buffer: 144 g glycine, 30 g Tris base/L of distilled water, pH 8.3) in distilled water containing 10 % methanol. The membrane was subsequently blocked for 30 min at room temperature in 5% w/v skimmed milk/PBS then incubated overnight at 4°C with the anti-bacterial peptidoglycan antibody (MAB 995 from Millipore, UK) diluted 1/500 in 5% w/v skimmed milk/PBS. Following 3 × 15 min washings in PBS containing 0.2% tween 20, the membrane was incubated in horse radish peroxidase (HRP)-conjugated goat anti-mouse Ig secondary antibody (Chemicon, Millipore, UK) diluted 1/10,000 in 5 % w/v skimmed milk/PBS for 2 h at room temperature. Following further washes in PBS/tween 20, (as above, 3 × 15 min each) spots were detected using the enhanced chemiluminescence detection reagent. Specific protein signal from the membranes was visualized using a ChemicDoc® (Bio-Rad, UK) and images captured with Image Lab® Software Version 3.0.1.
RESULTS
PCR and high throughput sequencing of brain samples
Ten out of 20 samples gave PCR products. The remaining 10 samples failed even after using a set of 14 primers in different combinations. Breaking the code showed that six (60%) of the positive samples were from non-AD participants, while four (40%) belonged to AD patients. A wide spectrum of bacteria was detected in samples from both groups, containing both oral and gastrointestinal tract microbiome species. The post-mortem delay for the AD specimens on average was 9 h. However, the storage interval from donation of brains to BDR and isolating DNA, for this study, on average, overall was 16 years. For the cases that were significantly associated with the presence of bacteria in AD, the average storage interval was 15 years (Table 1). The AD brain specimens without presence of bacteria were of slightly longer storage interval (17 years on average). The control, non-AD cases, on average had a longer post-mortem delay interval of 26 h compared with the AD cases with bacteria present. The storage interval with and without bacterial counts for the control, non-AD cases, on average, was 6 years. Whilst the cause of death for the AD cases with bacteria were unknown for 3 out of 4, one case resulted from heart failure (Table 1). For the control group, the cause of death was clearly defined (Table 1) whereby 3 out of 6 cases with bacterial counts had suffered from pneumonia, stroke and bacterial sepsis (multiple organ failure). Therefore, a significant difference between the AD and non-AD groups lies in the post-mortem interval and storage interval.
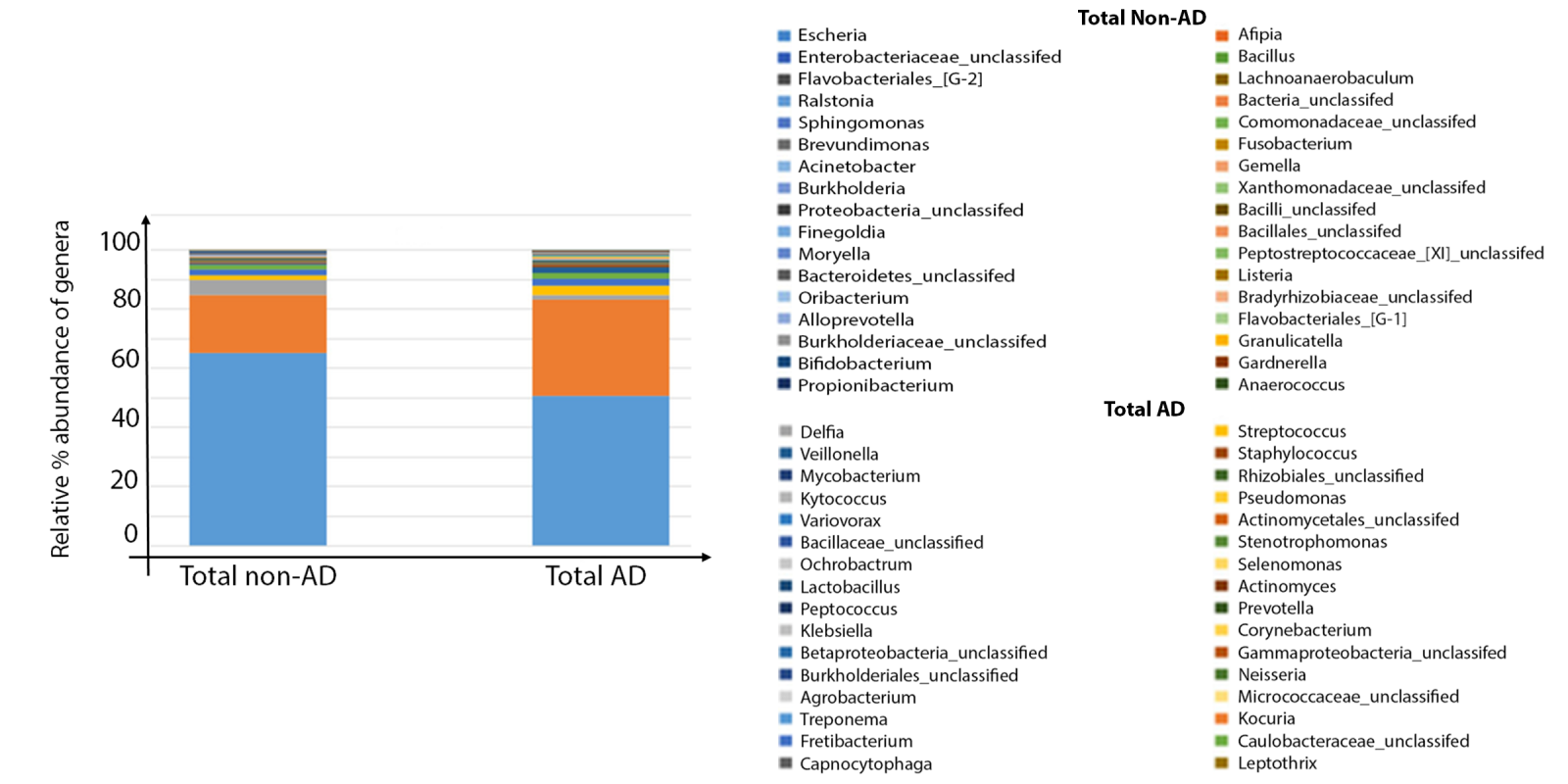
The distribution of bacterial taxa was sorted into two groups comprising of AD and non-AD (not shown). In the AD group the phylum Firmicutes was considerably higher, and the Proteobacteria phylum was much lower than in the non-AD group. The distribution of bacterial families was also determined and the dominating family in the AD group was Pseudomonadaceae. Figure 1 shows the relative amounts of a great number of genera discovered in both groups (see Table 1 for actual numbers of bacteria). It was noteworthy that members of the Actinomycetales order and Prevotella species (typically of Gingivitis) and Treponema and Veillonella species (periodontitis) were present in the AD group, but not in the non-AD group. Conversely, the genus Fusobacterium was seen in the non-AD group, but not in the AD group.
Figure 1: Bacterial reads compiled for the number of genera detected in non-AD (n=10) and AD (n=10) brains. The Y-axis represents relative percentage (%) abundance of bacterial genera.
Statistics for post-mortem delay and storage interval
The Shapiro-Wilk T-test for independent samples showed that the post-mortem interval was normally distributed, but that storage interval was not. Therefore, the non-parametric Mann-Whitney U test was used for independent samples for storage interval. In both cases the difference was highly significant (p-value=0001). Post-mortem interval mean: Shapiro-Wilk, T-test (independent samples) for AD=9 h and for the non-AD=26 h (p-value=0.0001). For storage interval mean: Mann-Whitney U test (independent samples) for AD=16 years and non-AD 6 years (p-value=0.0001).
Bacterial peptidoglycan dot blot for all brains
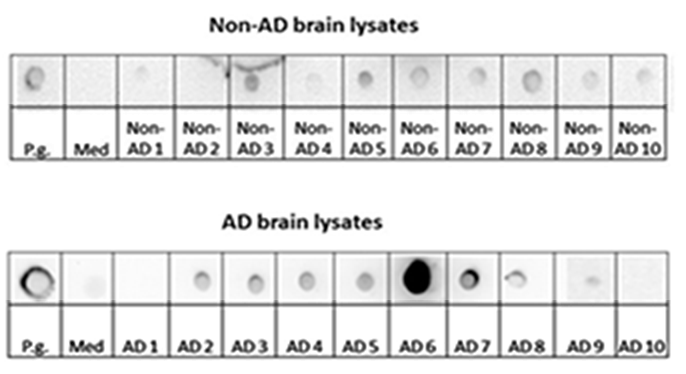
Dot blot analysis from tissue lysates from all the brains tested, confirmed the presence of the bacterial peptidoglycan protein. The positive control bacterial cell lysate from P. gingivalis (P.g.) was positive, and the sterile medium (Med) containing proteins used as a negative control remained negative for both groups of dot blots. Whilst non-AD brain lysates from case 2 and case 4 were negative, the non-AD (cases 1, 3, 5, 6, 7, 8, 9, 10) demonstrated unequivocal density around the specimen spots albeit with weaker density around each of the specimen spots. The AD brain lysates from the AD case 1 and AD case 10 brain lysates were negative whilst only a weaker spot appeared in AD brain lysate case 9 compared with AD (cases 2, 3, 4, 5, 6, 7, 8). The AD cases 6 and 7 brain lysates demonstrated a significantly greater density of the specimen dot spots (Figure 2).
Figure 2: Dot blot using lysates from non-AD (n=10) and AD (n=10) brains, bottled with mouse anti-bacterial peptidoglycan (MAB 995). The first dot in both blots represents lysate from P. gingivalis (P.g.) as a positive control for Gram-negative bacteria and the med refers to sterile medium used to culture (P.g.) as a source of irrelevant protein as negative control. Non-AD 1-10 are control brains showing some bacterial peptidoglycan. The AD 1-10 brain lysates when dot blotted demonstrated a significantly greater density in AD 6 suggesting a Gram positive bacterial contribution, whilst AD 3, 5, 8 and 10 that were positive for P. gingivalis LPS [28] were weak in their dot blot density.
DISCUSSION
Investigating microbial infections in the context of AD brains have a negative connotation because of the potential for cross-contamination of the brain tissue obtained at post-mortem with organisms from bowel or systemic tissue. Even if compelling evidence indicates a specific pathogen to contribute to the pathogenesis of AD, it is unlikely that a single “infectious” agent is the exclusive cause of this neurodegenerative disease.
It is noteworthy that individuals with the ApoE ε4 gene are vulnerable to greater amounts of Aβ, oxidative stress, lysosomal leakage, infections and pathogen/Aβ driven inflammation. These host stressors appear to have little effect on the survival of Actinomycetales, perhaps because of their association with multispecies of bacteria, which together (as a biofilm) tolerate adverse conditions [36].
With regards to post-mortem delay, this study demonstrated a significant difference in post-mortem delay in controls (26 h) compared with AD cases (9 h). However, the storage interval at -80ºC was much shorter than for control cases (6 years) compared with AD cases (16 years).
Within the Cohort, age range at death, and gender were similar, however, we cannot rule out whether or not the duration of tissue storage (16 years) had added to potential cross-contaminations in the AD cases, despite our results showing a minimal effect. Thus, the issue is more likely to be related to post-mortem interval and DNA degradation rather than cross-contamination.
Appreciation of the use of high throughput sequencing methodology is important in view of its likely future application in “the diagnostic standard worldwide”. It enables earlier discovery of novel infectious agents that may be involved in the development of complex diseases with unknown causes, which are currently difficult to detect using conventional procedures. In addition, the DNA from stored tissue can be examined retrospectively if the problem associated with degradation of DNA can be overcome.
In this study, electrophoresis of the PCR products demonstrated ten brain tissue specimens to show positive amplicons, while the remaining ten were negative. Six of the 10 positive PCR specimens belonged to non-AD participants, while the other four were from AD brains. This outcome does not necessarily mean that individual brains were free of bacteria, because DNA is known, under post-mortem conditions, to degrade rapidly. From this investigation, it appears that a time scale of post-mortem delay (9-26 h), and the storage interval at -80ºC (16-6 years) would require further refinements.
However, using 16S rDNA high throughput sequencing, this study detected a wide range of different bacterial taxa belonging to the phylogenetic groups Bacteroidetes (Treponema, Prevotella) and Actinomycetes, exclusively in tissue from four AD (Cases 2, 3, 8, 10) (Table 1). Sequencing analysis confirmed Actinomycetes exclusively in AD tissue compared with normal brains [31], and P. gingivalis LPS was identified in AD (Cases 3, 5, 8 and 10). Surprisingly AD case 6 (which demonstrated the most prominent dot blot signal) did not reveal bacterial DNA using the 16S rDNA high throughput sequencing or [32] using PCR methodology, suggesting that bacterial LPS may degrade more slowly than DNA.
Dot blot analysis for the bacterial peptidoglycan demonstrated bacteria to be present in at least eight brains in both (AD and non-AD) groups without significant difference. P. gingivalis LPS and the anti-peptidoglycan antibody did not recognize this bacterial outer membrane component, however, AD case 10 (Table 1) but, not case 1, provided evidence for bacterial content following PCR [9,32]. It is possible that P. gingivalis LPS from the outer membrane vesicles may have bound to other species of bacteria, particularly Actinomycetes; as demonstrated in AD cases 3 and 10 (Table1) [9,37]. In support of this finding, Actinobacteria are commonly found in the oral cavity and in the dysbiotic gastrointestinal microbiota of AD patients [10]. P. gingivalis outer membrane vesicles are known to assemble with Actinomyces viscosus cells [38]. Furthermore, the gene coding for a 40 kDa protein in the outer membrane vesicle protein of P. gingivalis has previously been cloned [39] and was confirmed as an important aggregation factor between P. gingivalis and A. viscosus [40]. The increased antibody levels to F. nucleatum and P. intermedia have been detected in AD patients [13] indicating the relevance of bacterial gingivitis/periodontitis and Actinobacteria in this study.
Understanding the AD microbiome is important, although this study shows P. gingivalis DNA was not recovered from either AD cases or from non-AD-cases. One explanation is the scarcity of P. gingivalis DNA in the brain samples, as this bacterium is found in low copy numbers, even in its primary oral niche (periodontal pocket), where it is a keystone bacterium of tissue destruction [41,42]. In addition, due to the poor quality of DNA extracted from end-stage AD brains at post-mortem, DNA from P. gingivalis may not have been amplified. Although not all bacteria were affected, P. gingivalis may be an exception in sensitivity to PCR inhibitors and other reagents that are used during nucleic acid extraction or copurified components from the biological sample such as bile salts, urea, haeme, heparin, and immunoglobulin Ig [43,44]. This limitation was identified and addressed in a former study; here the same tissue specimens were analyzed with biochemical and immunofluorescence methodology, detecting LPS as a signature of P. gingivalis in AD but not control cases (4/10) [9]. In this study, oral and intestinal bacterial DNA were also identified in the brain tissue from both AD and non-AD subjects using DNA and high throughput sequencing methodology.
CONCLUSION
In conclusion, the high throughput sequencing methodology is likely to be integrated into medical diagnostics worldwide and therefore there is a need to test its application for stored brain tissues. Our study demonstrated its applicability is plausible.It remains unclear whether or not bacteria within the post-mortem AD brain are related to systemic bacteria associated with the causes of death in AD cases, or to changes in bacterial populations following death. This study, nevertheless, confirms the presence and identification of bacterial species associated with gingivitis and periodontal disease in the AD brains, supporting former (prospective and retrospective) laboratory based and epidemiological studies. Furthermore, this study highlights Actinobacteria possibly binding with P. gingivalis LPS and this has relevance for AD pathophysiology. We do not suggest that oral bacteria (gingivitis and/or periodontitis pathogens) are responsible for all AD cases, but poor oral hygiene, which promotes bacterial related periodontal disease, may be a modifiable risk for AD in some individuals.
Conflict of Interest
There is no conflict of interest in the present study for any of the authors. Funding was as given under Acknowledgments.
Acknowledgments
The authors thank Brains for Dementia Research (BDR) and the Newcastle Brain Tissue Resource, UK for the human brain specimens and their invaluable help and support on ad hoc basis. The Newcastle Brain Tissue Resource is supported by the UK Medical Research Council, Alzheimer’s Research UK, and the Alzheimer’s Society through the Brains for Dementia Research Initiative and by the National Institute for Health Research (NIHR) Newcastle Biomedical Research Centre based at the Newcastle upon Tyne Hospitals NHS Foundation Trust and Newcastle University. SKS wishes to thank the University of Central Lancashire and the PreVisor awards (2017 and 2019) for their financial support. IO, HS and ERKE are grateful for funding through the European Commission (FP7-HEALTH-306029 “TRIGGER”). The Department of Biochemistry DNA Sequencing Facility of the University of Cambridge, UK is acknowledged for sequencing. Mr. Torbjørn K Aaberg is thanked for help with Figure 1.
REFERENCES
- Hu X, Wang T, Jin F (2016) Alzheimer‘s disease and gut microbiota. Sci China Life Sci 59(10): 1006-1023.
- Olsen I, Singhrao SK (2015) Can oral infection be a risk factor for Alzheimer’s disease? J Oral Microbiol 7: 29143.
- Pritchard AB, Crean S, Olsen I, Singhrao SK (2017) Periodontitis, microbiomes and their role in Alzheimer’s disease. Front Aging Neurosci 9: 336.
- Corder EH, Saunders AM, Strittmatter WJ, Schmechel DE, Gaskell PC, Small GW, et al. (1993) Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. Science 261(5123): 921-923.
- Harris JD, Evans V, Owen JS (2006) ApoE gene therapy to treat hyperlipidemia and atherosclerosis. Curr Opin Mol Ther 8(4): 275-287.
- Moretti EW, Morris RW, Podgoreanu M, Schwinn DA, Newman MF, Bennett E, et al. (2005) ApoE polymorphism is associated with risk of severe sepsis in surgical patients. Crit Care Med 33(11): 2521-2526.
- Watts A, Crimmins EM, Gatz M (2008) Inflammation as a potential mediator for the association between periodontal disease and Alzheimer’s disease. Neuropsychiatr Dis Treat 4(5): 865-876.
- Tsoi LM, Wong KY, Liu YM, Ho YY (2007) Apoprotein E isoform-dependent expression and secretion of pro-inflammatory cytokines TNF-alpha and IL-6 in macrophages. Arch Biochem Biophys 460(1): 33-40.
- Poole S, Singhrao SK, Kesavalu L, Curtis MA, Crean S (2013) Determining the presence of periodontopathic virulence factors in short-term postmortem Alzheimer’s disease brain tissue. J Alzheimers Dis 36(4): 665-677.
- Zhuang ZQ, Shen LL, Li WW, Fu X, Zeng F, Gui L, et al. (2018) Gut microbiota is altered in patients with Alzheimer's Disease. J Alzheimers Dis 63(4): 1337-1346.
- Tzeng NS, Chung CH, Yeh CB, Huang RY, Yuh DY, Huang SY, et al. (2016) Are chronic periodontitis and gingivitis associated with dementia? A nationwide, retrospective, MATCHED-Cohort study in Taiwan. Neuroepidemiology 47(2): 82-93.
- Chen C-K, Wu Y-T, Chang Y-C (2017) Association between chronic periodontitis and the risk of Alzheimer’s disease: A retrospective, population-based, matched-cohort study. Alzheimers Res Ther 9: 56.
- Sparks Stein P, Steffen MJ, Smith C, Jicha G, Ebersole JL, Abner E, et al. (2012) Serum antibodies to periodontal pathogens are a risk factor for Alzheimer's disease. Alzheimers Dement 8(3): 196-203.
- Stein PS, Desrosiers M, Donegan SJ, Yepes JF, Kryscio RJ (2007) Tooth loss, dementia and neuropathology in the Nun Study. J Am Dent Assoc 138(10): 1314-1322.
- Yin C, Ackermann S, Ma Z, Mohanta SK, Zhang C, Li Y, et al. (2019) ApoE attenuates unresolvable inflammation by complex formation with activated C1q. Nat Med 25(3): 496-506.
- International Genomics of Alzheimer’s Disease Consortium (IGAP) (2015) Convergent genetic and expression data implicate immunity in Alzheimer’s disease. Alzheimers Dement11(6): 658-671.
- Singhrao SK, Olsen I (2018) Assessing the role of Porphyromonas gingivalis in periodontitis to determine a causative relationship with Alzheimer's disease. J Oral Microbiol 11(1): 1563405.
- Montagne A, Barnes SR, Sweeney MD, Halliday MR, Sagare AP, Zhao Z, et al. (2015) Blood-brain barrier breakdown in the aging human hippocampus. Neuron 85(2): 296-302.
- Vitek MP, Brown CM, Colton CA (2009)ApoE genotype-specific differences in the innate immune system. Neurobiol Aging 30(9): 1350-1360.
- Balin BJ, Gerard HC, Arking EJ, Appelt DM, Branigan PJ, Abrams JT, et al. (1998) Identification and localization of Chlamydia pneumoniae in the Alzheimer’s brain. Med Microbiol Immunol 187(1): 23-42.
- Gerard HC, Wildt KL, Whittum-Hudson JA, Lai Z, Agerand J, Hudson AP (2005) The load of Chlamydia pneumoniae in the Alzheimer’s brain varies with ApoE genotype. Microbial Pathogenesis, 39(1-2): 19-26.
- Itzhaki RF, Wozniak MA (2006) Herpes simplex virus type 1, apolipoprotein E and cholesterol: A dangerous liaison in Alzheimer’s disease and other disorders. Prog Lipid Res 45(1): 73-90.
- Gérard HC, Wang GF, Balin BJ, Schumacher HR, Hudson AP (1999) Frequency of apolipoprotein E (ApoE) allele types in patients with Chlamydia-associated arthritis and other arthritides. Microb Pathog 26(1): 35-43.
- MacIntyre A, Abramov R, Hammond CJ, Hudson AP, Arking EJ, Little CS, et al. (2003) Chlamydia pneumoniae infection promotes the transmigration of monocytes through human brain endothelial cells. J Neurosci Res 71(5): 740-750.
- Urosevic N, Martins RN (2008) Infection and Alzheimer's disease: The ApoE epsilon4 connection and lipid metabolism. J Alzheimers Dis 13(4): 421-435.
- Itzhaki RF, Wozniak MA (2010) Alzheimer's disease and infection: Do infectious agents contribute to progression of Alzheimer's disease? Alzheimers Dement 6(1): 83-84.
- Dominy SS, Lynch C, Ermini F, Benedyk M, Marczyk A, Konradi A, et al. (2019) Porphyromonas gingivalis in Alzheimer's disease brains: Evidence for disease causation and treatment with small-molecule inhibitors. Sci 5(1): eaau3333.
- Miklossy J (2011) Alzheimer’s disease: A neurospirochetosis. Analysis of the evidence following Koch’s and Hill’s criteria. J Neuroinflammation 8: 90.
- Miklossy J (2011) Emerging roles of pathogens in Alzheimer disease. Expert Rev Mol Med 13: e30.
- Riviere GR, Riviere KH, Smith KS (2002) Molecular and immunological evidence of oral Treponema in the human brain and their association with Alzheimer’s disease. Oral Microbiol Immunol 17(2): 113-118.
- Emery DC, Shoemark DK, Batstone TE, Waterfall CM, Coghill JA, Cerajewska TL (2017) 16S rRNA next generation sequencing analysis shows bacteria in Alzheimer’s post-mortem brain. Front Aging Neurosci 9: 195.
- Poole S (2014) Aetiological links between oral pathogens and dementia. PhD Thesis. Awarded by the University of Central Lancashire.
- Kozich JJ, Westcott SL, Baxter NT, Highlander SK, Schloss PD (2013) Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the MiSeq Illumina sequencing platform. Appl Environ Microbiol 79(17): 5112-5120.
- Siddiqui H, Chen T, Aliko A, Mydel PM, Jonsson R, Olsen I (2016) Microbiological and bioinformatics analysis of primary Sjögren’s syndrome patients with normal salivation. J Oral Microbiol 8: 31119.
- White JR, Nagarajan N, Pop M (2009) Statistical methods for detecting differentially abundant features in clinical metanogenic samples. PLoS Comput Biol 5(4): e1000352.
- Brändle N, Zehnder M, Weiger R, Waltimo T (2008) Impact of growth conditions on susceptibility of five microbial species to alkaline stress. J Endod 34(5): 579-582.
- Singhrao SK, Olsen I (2018) Are Porphyromonas gingivalis outer membrane vesicles microbullets for sporadic Alzheimer's disease manifestation? J Alzheimers Dis Rep 2(1):219-228.
- Ellen RP, Grove DA (1989) Bacteroides gingivalis vesicles bind to and aggregate Actinomyces viscosus. Infect Immun 57(5): 1618–1620.
- Abiko Y, Hayakawa M, Aoki H, Kikuchi T, Shimatake H, Takiguchi H (1990) Cloning of a Bacteroides gingivalis outer membrane protein gene in Escherichia coli. Arch Oral Biol 35(9): 689-695.
- Hiratsuka K, Abiko Y, Hayakawa M, Ito T, Sasahara H, Takiguchi H, et al. (1992) Role of Porphyromonas gingivalis 40-kDa outer membrane protein in the aggregation of P. gingivalis vesicles and Actinomyces viscosus Arch Oral Biol 37(9): 717-724.
- Hajishengallis G, Darveau RP, Curtis MA (2012) The keystone-pathogen hypothesis. Nature Rev Microbiol 10(10): 717-725.
- Darveau RP, Hajishengallis G, Curtis MA (2012) Porphyromonas gingivalis as a potential community activist for disease. J Dent Res 91(9): 816-820.
- Nolan T, Hands RE, Ogunkolade W, Bustin SA (2006) SPUD: A quantitative PCR assay for the detection of inhibitors in nucleic acid preparations. Anal Biochem 351(2): 308-310.
- Sundquist A, Bigdeli S, Jalili R, Druzin ML, Waller S, et al. (2007) Bacterial flora typing with deep, targeted, chip-based pyrosequencing. BMC Microbiol 7: 108.
Copyright: Siddiqui H, et al. ©2019. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Siddiqui H (2019). High Throughput Sequencing Detect Gingivitis and Periodontal Oral Bacteria in Alzheimer’s Disease Autopsy Brains. Neuro Research 1(1): 3.
 Abstract
Abstract  PDF
PDF